
As discussed in CIBSE’s comprehensive guide TM13,1 many people are exposed to legionella , but their bodies’ defence systems respond to prevent illness. Although previously healthy people may develop Legionnaires’ disease (LD), there are several factors that have been shown to increase an individual’s susceptibility, including: increasing age; being male; being a smoker; having an existing respiratory disease; and pre-existing illness, such as cancer, diabetes, kidney disease or alcoholism.
In a particularly prescient review paper published in 2019, Bradley2 et al note that ‘lower respiratory infections remain one of the top global causes of death and the emergence of new diseases continues to be a concern. In the first two decades of the 21st century, we have borne witness to the emergence of newly recognised coronaviruses that have rapidly spread around the globe….
Although we perhaps most frequently think of viruses when discussing emerging respiratory infections, bacteria have not been left out of the mix, as we have witnessed an increase in the number of infections from Legionella’. European data reported3 by Cunha et al suggests that LD is substantially under-diagnosed and under-reported.
Comparing 2011 data of reported LD from the European Legionnaires’ Disease Surveillance Network with that of a German multicentre study that employed active testing across a population indicates that possibly fewer than 5% of cases are being reported. If extrapolated, this would infer – based on 2018 EU data4 – that, rather than the 20 reports of LD per million Europeans, there could be more than 400 cases per million.
In the UK, there has been a steady rise in reported cases – in 2019 there were 516 confirmed cases in England and Wales5 (approximately 8.5 per million), and this is likely to be significantly fewer than actual cases. As with all potentially fatal health problems, there are associated personal tragedies, as well as significant financial consequences6,7 for the building operator if there is a failure to control the risk to occupants properly.
As described by AWT,8 Legionella pneumophila was the first legionella species, named upon discovery in 1977 after the 1976 disease outbreak in Philadelphia. It was determined that neither the bacterium nor the disease was new, and that legionella bacteria have been around and causing disease for many years; it is found in the ground and groundwater.
Legionella pneumophila is considered as the single species that is responsible for more than 80% of legionellosis cases. Outbreaks of LD have principally been associated with specific variations (or ‘serotypes’) collectively identified as Legionella pneumophila serogroup 1 – the most common legionella in environmental samples.
Legionellosis notably includes LD, a severe pneumonia infection, and Pontiac fever, an acute flu-like illness. Even though the colonisation of a water system by legionella may occur frequently, this alone is not enough to pose a high risk to humans, unless the bacteria population reaches high numbers and becomes dispersed through appropriate aerosolisation to impact susceptible people by inhalation.5
Legionella has evolved to exist and multiply in adverse conditions by being a parasite. (The boxout, ‘Prozotoa and amoebae’, provides some associated terminology.) Scheid9 explains that the term ‘free-living amoeba’ (FLA) is applied to a mixed group of parasitic amoebae that are found in biofilms (which can visually manifest as slime), which are a complex layer of microorganisms that have attached and grown on a surface.
Some FLAs have a resistant life stage, a cyst, which largely protects the organism from adverse environmental conditions – such as higher temperatures and chemical disinfectants – and, therefore, provides great tenacity. The FLA feed on algae, bacteria and fungi, as well as on smaller protozoa, and provide protected hosts for bacteria.
The FLAs Acanthamoeba and Vermamoeba vermiformis are microscopic, free-living amoebae that exist in water and soil and are considered10 suitable hosts for pathogenic microorganisms (bacteria that can cause disease). Work by Rowbotham11 identified that these FLA can serve as hosts, reservoirs, vehicles and protectors for legionella (Figure 1).
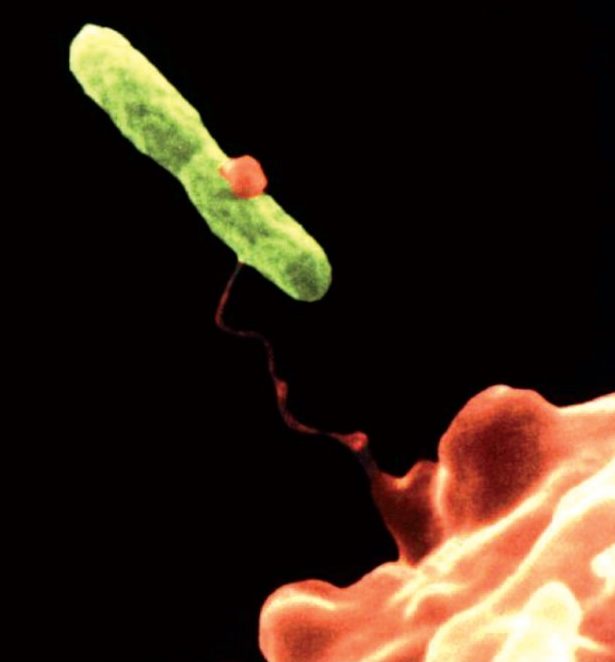
Figure 1= Vermamoeba vermiformis, orange, entraps Legionella pneumophila bacteria (≈2 to 20μm long), green (Source: pixnio.com/science/microscopy-images/hartmannella-vermiformis)
A recent systematic literature review12 by Nisar et al shows that Legionella pneumophila combined with Acanthamoeba and Vermamoeba were extensively found in chlorinated and thermally treated water, which indicated the potential tolerance of Legionella pneumophila and its protozoan hosts to survive under a wide range of disinfection conditions.
Not only does legionella acquire necessary nutrients from its host, but the host also protects legionella from toxic and unfavourable environmental conditions. AWT1 notes that disinfection of a water source can lead to the eradication or reduction of legionella populations; however, the majority of legionella does not exist in an independent and solitary state (like a plankton). Instead, legionella more often resides inside a protozoan host and/or biofilm community.
When legionella is released from a host, it is encapsulated within vesicles (small fluid-filled bladders) derived from the host’s cell membrane. There can be more than a thousand legionella within a single vesicle, and legionella dispersed within these vesicles appears to be more virulent than legionella remaining parasitised in a protozoan host.
There are several authoritative guides to controlling and managing legionella risk in the built environment, including: CIBSE TM13:2013; the newly revised ASHRAE Guideline 12-2020; Health Technical Memorandum 04-01:2016; and the recently updated BS 8580-1:2019 Water quality – Risk assessments for Legionella control – Code of practice, which includes significant additions including an increased emphasis on risk assessment and management. These all provide further references to what is an increasingly well-documented topic.
The freely accessible Health and Safety Executive’s (HSE’s) approved code of practice, Legionnaires’ disease: The control of legionella bacteria in water systems (L8), 2013, contains practical guidance on how to manage and control the risks in building systems – see www.hse.gov.uk/legionnaires.
Part 2 of the complementary technical guidance in HSG274 relates specifically to hot and cold water systems. Practically, temperature control is the traditional strategy for reducing the risk of legionella in hot and cold water systems, as temperatures between 20°C and 45°C have been identified as the optimal range for legionella growth. HSG274 specifically notes that, if hot water is stored, it should be at least at 60°C and distributed so that it reaches a temperature of 50°C (55°C in healthcare premises) within one minute at the outlets.
The WRAS information leaflet Preheated domestic hot water – Storage of preheated domestic hot water and possible growth of legionella bacteria notes that legionella bacteria in concentrations of 100,000 colony forming units per litre (cfu.L-1, a measure of viable bacterial cells) and higher are not uncommon at the base of conventional hot water storage vessels where temperatures of 20°C to 45°C are maintained.
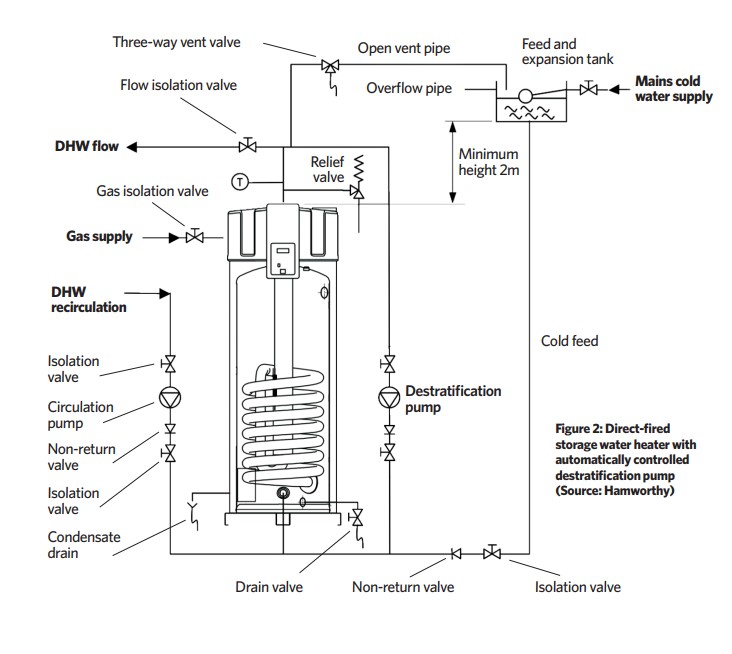
For comparison, HSG274 Part 2 recommends review of control measures (and possible disinfection) where legionella concentrations are in the range 100 to 1,000cfu.L-1, and corrective action (followed by retesting every few days) where they exceed 1,000cfu.L-1. An example of simple, practical, system design to reduce the risk of legionella accumulation is shown in Figure 2, where a direct-fired storage water heater installation includes a destratification pump that is automatically controlled to provide a daily legionella protection cycle, by circulating water from top to bottom in the cylinder of water at 60°C.
Coincidentally, the main wholesome hot water (DHW) recirculation pump can be activated to provide a circulation of hot water around the distribution circuit. Although this should be undertaken at times when occupants will not be drawing hot water – as water at 60°C presents a scalding risk – local thermostatic mixing valves should be provided if this is considered an unacceptable risk. (TMVs will, themselves, present further requirements for legionella management.)
In the recent review paper13 by Whiley, H et al, they reported that up to 25% of Legionella pneumophila cells can survive heat treatment of 70°C, but all of these were in a viable, but non-culturable, state. This could be significant in future assessments of legionella control, as being ‘non-culturable’ means it will not grow on the growth media that are used in culture methods currently employed to determine the efficacy of disinfection protocols.
In older, more complex buildings, it may be practically impossible to maintain the required temperatures. In the coverage14 of legionella protection by the US Centers for Disease Control and Prevention (CDC), it highlights that ‘a building wholesome water system with extensive dead-legs, low disinfectant residuals, tepid hot water temperatures, minimal water flow, and an established legionella biofilm might promote substantial legionella growth and dissemination in weeks or months’.
So, in addition to maintaining a temperature-control regimen, there may be occasions where additional, properly managed biocidal treatment is required for the effective control of legionella. HTM 04 notes that ‘effective concentrations of some biocides are difficult to achieve in hot-water systems due to gassing off’.
HTM 04 raises a further issue that has gained increasing focus during the control period for Covid-19, on how the use of antimicrobial hand-rubs might impact on the frequency of use of wash-hand basins and the volume of water being distributed, leading to the increased potential of stagnancy and low-water temperatures that supply wash-hand basins.
Where biocides are used to control microbial growth in water systems, as with the temperature regimen, meticulous control and monitoring programmes should be in place if they are to be effective.
The review12 of studies undertaken by Nisar indicates that there was a failure of common disinfection procedures to achieve long-term elimination of Legionella pneumophila and protozoan hosts from potable (wholesome) water, and that disinfection procedures and protozoan hosts may facilitate the reproduction of viable Legionella pneumophila that have been shown to be highly resistant to many water disinfection protocols.
In an article15 by Scherberger, he notes that biofilms are not simply removed as ‘biofilm is produced to protect bacteria from harsh environments and disinfectants such as bleach, and antibiotics fall into the definition of a harsh environment’.
Removal from water systems will probably require a carefully managed process with specialist advice, chemicals and appropriate personal protective equipment (PPE), so as not to adversely affect the operatives or the building occupants. Novel methods have been reported by Migliarina16 that use hypochlorous acid, which reportedly provides a safe and effective method to remove biofilms in existing piped wholesome water systems.
Recently, Cloutman-Green et al17 reported on a longitudinal study of a copper and silver ionisation system that, following a specific derogation of standing rules, was employed in a London clinical building with a wholesome hot-water system – both storage and supply network – operated nominally at 43°C, which obviated the risk of scalding in this healthcare setting.
Over a six-year period, the systems were comprehensively sampled and, throughout that period, outlet temperatures varied between 37°C and 42°C, but there was no detection of any Legionella pneumophila.
The copper and silver levels were maintained at target levels of >0.2mg.L-1 and >0.02mg.L-1, respectively. The energy savings and reduction of carbon emissions were calculated to amount to 33% and 24%, respectively, compared with an equivalent temperature-controlled system. Such techniques require careful monitoring and control to ensure adequate dosing.
There are clearly practical measures that can be taken to reduce the risks from legionella in wholesome hot-water systems, but, as with other risks to building occupants, continuous assessment and management are the order of the day.
Protozoa and amoebae
The term ‘proto-zoa’ literally means ‘first animals’, and originally connected as the basis of animal evolution. However, they were subsequently recognised as a discrete group on the basis of being a single-celled (unicellular) organism, and were assigned to the collective name of protozoa, although the taxonomy is still developing as research continues. Protozoa have a nucleus surrounded by a membrane (eukaryotic organisms), which exist as structurally and functionally independent individual cells, including those that form colonies. Protozoa have developed relatively complex subcellular features (membranes and special structures) that enable them to survive environmental rigours. Most protozoa are microscopic organisms – only a few grow to a size large enough to be visible to the naked eye. As unicellular eukaryotes, they move about to survive, feed and breed.
Amoebae are one of four main groups of protozoa, and use temporary, thread-like or balloon-like extensions of the cell membrane – known as pseudopodia (‘false feet’) – to creep or crawl over solid substrates.
■ Much of this boxout is based on abbreviated information from parasite.org.au/para-site/contents/protozoa-intoduction.html
Recommissioning hot water systems after a period of shutdown
The methods will be dependent on the particular application. The article ‘Building closures could lead to legionella outbreak’ in June’s CIBSE Journal provides an overview of guidance for maintenance during the shutdown period. On the dedicated HSE web page,18 ‘Legionella risks during the coronavirus outbreak’ recommends that if hot-water outlets have not been flushed at least weekly to prevent water stagnation, they should be inspected by a competent person and, as required, cleaned and disinfected before the building is occupied.
The Legionella Control Association webpage19 gives more detail of the precautions and processes that will be required, as does the US-based CDC ‘8 steps to minimise legionella risk before your business or building reopens’.20
© Tim Dwyer, 2020.
References:
1 TM13:2013 – Minimising the risk of Legionnaires’ disease, CIBSE 2013
2 Bradley, B T et al, Emerging respiratory infections: the infectious disease pathology of SARS, MERS, pandemic influenza, and Legionella, Seminars in Diagnostic Pathology 36, 152–159, 2019 –
3 Cunha, B A et al, Legionnaires’ disease, Lancet 387, pp 376-385, 2016
4 European Legionnaires’ Disease Surveillance Network
5 Monthly Legionella Report, Public Health England, December 2019
6 HSE Royal United Hospital case, – accessed 1 June 2020
7 This Week in FM, – accessed 1 June 2020
8 AWT – Legionella 2019: A position statement and guidance document
9 Scheid, P, Relevance of free-living amoebae as hosts for phylogenetically diverse microorganisms, Parasitology Research 113, 2,407–2,414 (2014) –
10 Delafont, V et al, Vermamoeba vermiformis: a free-living amoeba of interest, Review, Microbial Ecology 2018 Nov, 76(4):991-1001, doi:10.1007/s00248-018-1199-8
11 Rowbotham, T, Preliminary report on the pathogenicity of Legionella pneumophila for freshwater and soil amoebae, Journal of Clinical Pathology 33:1179–1183, 1980
12 Nisar, M A, Legionella pneumophila and protozoan hosts: implications for the control of hospital and potable water systems
13 Whiley, H et al, Legionella persistence in manufactured water systems: pasteurization potentially selecting for thermal tolerance, Frontiers in Microbiology, 2017
14 Guidance for reopening buildings after prolonged shutdown or reduced operation, CDC, – accessed 1 June 2020
15 Scherberger, J, Identifying and eradicating biofilm (with HOCL), – accessed 1 June 2020
16 Migliarina, F et al, A modern approach to disinfection, as old as the evolution of vertebrates, Healthcare 2014; 2(4):516-526. doi:10.3390/healthcare2040516
17 Cloutman-Green, E et al, Controlling Legionella pneumophila in water systems at reduced hot-water temperatures with copper and silver ionization, AJIC 47 (2019) 761-766
18 Legionella risks during the coronavirus outbreak, HSE
19 Safe management of water systems in buildings in Covid-19,
20 Guidance for Reopening Buildings…, CDC
